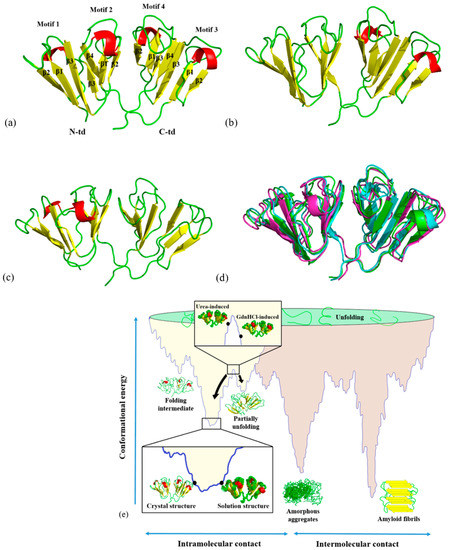
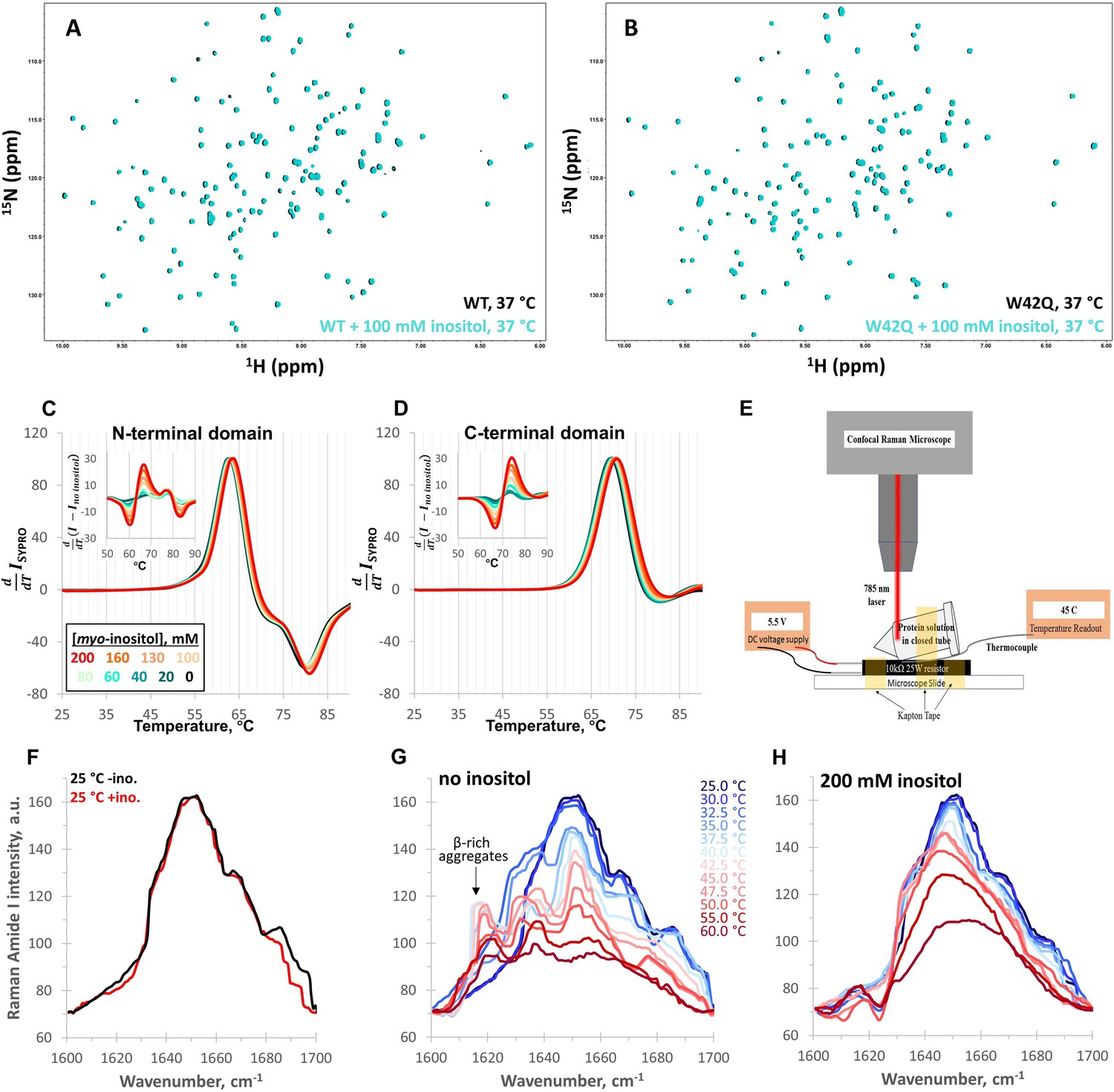
An alternative structural isoform in amyloid‐like aggregates formed from thermally denatured human γD‐crystallin - Moran - 2014 - Protein Science - Wiley Online Library

Plants | Free Full-Text | Constitutive and Adaptive Traits of Environmental Stress Tolerance in the Threatened Halophyte Limonium angustebracteatum Erben (Plumbaginaceae) | HTML

Cumulative deamidations of the major lens protein γS‐crystallin increase its aggregation during unfolding and oxidation - Vetter - 2020 - Protein Science - Wiley Online Library

Annales des Sciences Naturelles. Zoologia; biologia. 328 EUG. DAOAY DE DEES supra processu aculeiformi Yalidiusculo, antrorsumvergente; in parte qiiarta marginis interioris carina rotundata parum pro- miiiente et prope apicem iii margine
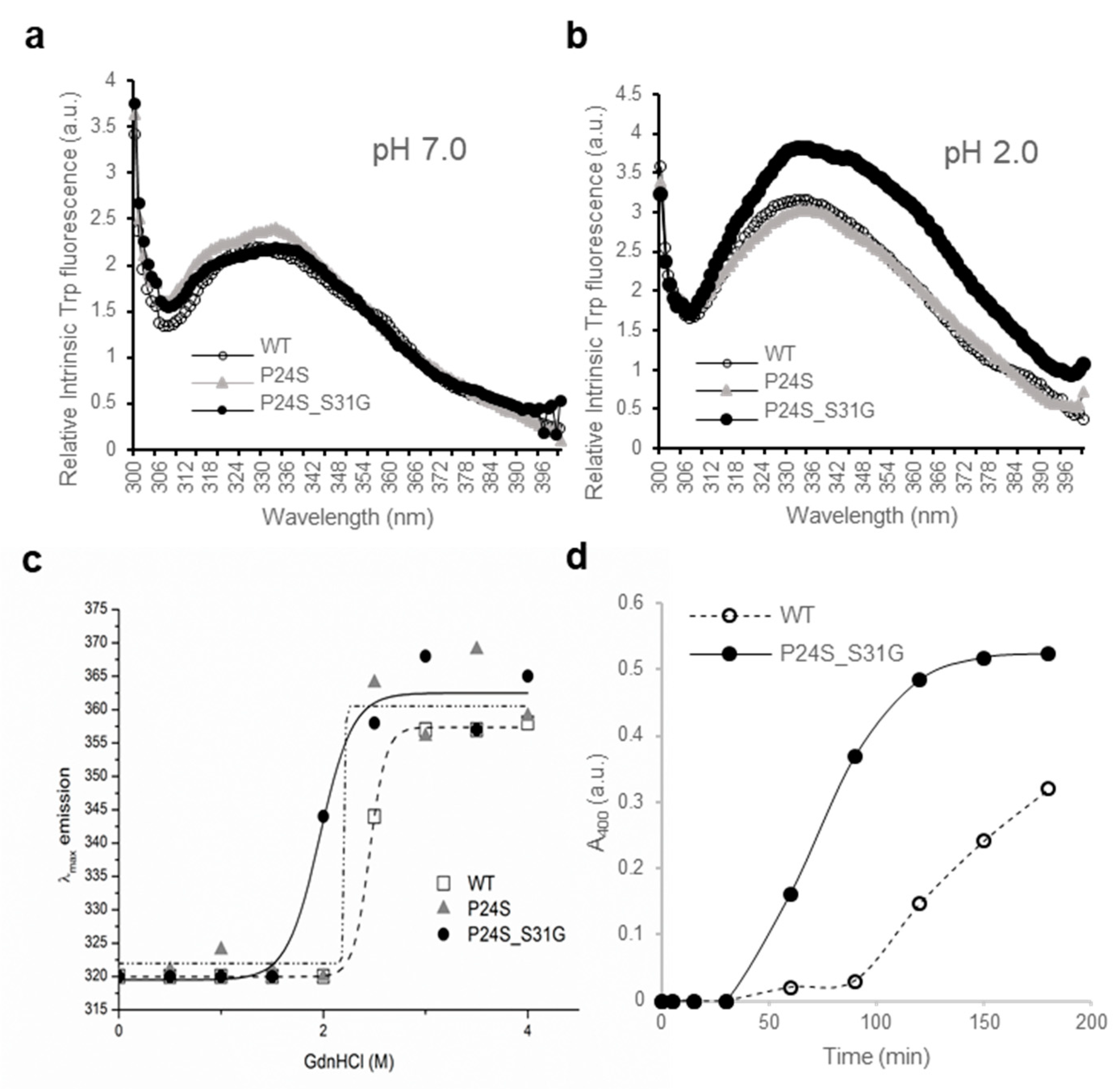
IJMS | Free Full-Text | Cataract-Associated New Mutants S175G/H181Q of βΒ2-Crystallin and P24S/S31G of γD-Crystallin Are Involved in Protein Aggregation by Structural Changes | HTML

Using Schematic Models to Understand the Microscopic Basis for Inverted Solubility in γD-Crystallin | The Journal of Physical Chemistry B

Crystal structure of the cataract‐causing P23T γD‐crystallin mutant - Ji - 2013 - Proteins: Structure, Function, and Bioinformatics - Wiley Online Library

PDF) Contributions to the characterization of Plantago species from Romania. Review | Nicoleta Ianovici - Academia.edu

Role of Conformational Flexibility in Monte Carlo Simulations of Many-Protein Systems | Journal of Chemical Theory and Computation